Misleading: US health agency did not find Lianhua Qingwen effective in COVID-19 treatment
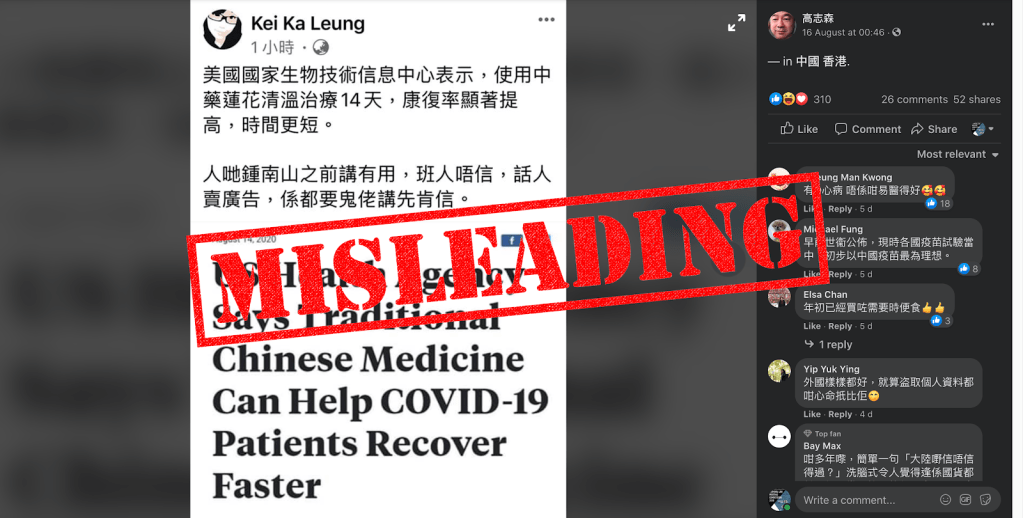
On Aug. 14, a news article in the Philippines claimed that the U.S. National Center for Biotechnology Information (NCBI) found Lianhua Qingwen (LH) capsules (连花清瘟) made from traditional Chinese herbal medicine to be effective in speeding up recovery from COVID-19.
Citing a research paper published in May on PubMed Central, an online archive of biomedical journals managed by the NCBI, the report said NCBI’s study discovered that a 14-day use of LH capsules achieved “a significantly higher rate of, and a shorter time to, symptom recovery” among COVID-19 patients.
The article was shared by Hong Kong and mainland China Facebook users including Ko Chi-Sum, an outspoken film producer with more than 104,000 followers on the social media platform.
His post received more than 300 likes and 50 shares. Di Bar (帝吧), a well-known pro-government Chinese group, also shared the article on their Facebook group page with almost 76,000 members.
The claim is misleading. The study in question was actually conducted by researchers in China, including Zhong Nanshan, leader of China’s National Health Commission’s COVID-19 research group. The paper was accepted by a journal called Phytomedicine, which is published by a Dutch company, Elsevier.
NCBI neither conducted the research nor endorsed the results. It simply manages the online medical journal archive that collects “articles from thousands of journals,” according to its website.
As a disclaimer, PubMed Central emphasizes that it is not a publisher. It relies on the scientific publishing process of each journal to identify and address problems. It also states that the views and opinions of authors expressed on PubMed Central do not reflect those of the U.S. government.
The findings of the study seem to have been misinterpreted as well. While LH capsules were found to facilitate recovery from COVID-19, the drug only has therapeutic effects and is not a cure for the disease.
The research shows LH capsules can alleviate mild symptoms such as fever, coughing and fatigue. It also points out the drug had no effect in preventing more severe conditions or reducing the viral load among 284 tested COVID-19 patients.
In mainland China, LH capsules were added to standard COVID-19 therapy in February.
But beyond its home country, the use of the drug as medication for COVID-19 patients remains limited. In the Philippines, the director general of the Food and Drug Administration (FDA), Eric Domingo, clarified on Aug. 13 that LH capsules have only been endorsed as treatment for fever, muscle soreness and other symptoms, but not as a treatment for COVID-19.
In the U.S., LH capsules have not been given the green light as a product that can “mitigate, prevent, treat, diagnose, or cure COVID-19.” A marketing website called lianhuaqingwencaps.com received a warning letter from the U.S. FDA on July 6 and has since shut down.


